Polyolefin in Pharmaceutical Packaging Market 2026–2036: Global Industry to Reach USD 36,001.5 Mn by 2036 at 4.6% CAGR
Polyolefin demand in Brazil’s pharma packaging to grow at 5.3% CAGR by 2036, driven by ANVISA regulations and public healthcare boosting compliant polymer use.
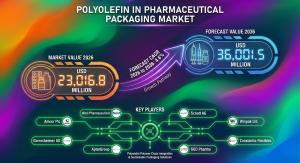
NEWARK, DE, UNITED STATES, February 26, 2026 /EINPresswire.com/ -- The global polyolefin in pharmaceutical packaging market is set to witness steady, regulation-driven growth as pharmaceutical production scales globally and packaging requirements become increasingly stringent. According to a newly released study by Future Market Insights (FMI), the market was valued at USD 21,839.3 million in 2025 and is projected to reach USD 23,016.8 million in 2026, before expanding to USD 36,001.5 million by 2036, registering a compound annual growth rate (CAGR) of 4.6% during the forecast period.
The report highlights that the market is expected to generate an absolute dollar opportunity of USD 12,984.7 million between 2026 and 2036, reflecting consistent demand for high-performance, compliant packaging materials across pharmaceutical supply chains. Growth is underpinned by rising global drug manufacturing, increasing adoption of biologics, and a strong shift toward advanced primary packaging systems designed to ensure product stability and regulatory compliance.
Key Market Highlights
• Market Value (2025): USD 21,839.3 million
• Market Value (2026): USD 23,016.8 million
• Forecast Value (2036): USD 36,001.5 million
• CAGR (2026–2036): 4.6%
• Absolute Dollar Growth: USD 12,984.7 million
Discover Growth Opportunities in the Market – Get Your Sample Report Now
https://www.futuremarketinsights.com/reports/sample/rep-gb-32099
Market Overview: What Is Driving Growth?
Polyolefin materials including polyethylene (PE), polypropylene (PP), and specialty olefin-based polymers, are widely used in pharmaceutical packaging due to their chemical resistance, moisture barrier performance, lightweight structure, and compatibility with sterilization processes. These materials play a critical role in protecting drug formulations from contamination, degradation, and environmental exposure.
Demand is particularly strong across rigid packaging formats such as bottles, closures, vials, and syringe components, as well as flexible formats including blister packs, strip packs, and laminates. The increasing complexity of pharmaceutical formulations, especially biologics and injectables, is driving the need for high-purity, extractables-controlled polyolefin grades, including advanced materials such as COC/COP and metallocene-based polymers.
Demand Drivers: Regulation, Biologics, and Global Drug Production
1. Rising Pharmaceutical Production
The growth of solid oral and liquid drug formulations is increasing demand for HDPE bottles, closures, and blister packaging components. Global pharmaceutical output, particularly in emerging markets, continues to drive high-volume consumption of polyolefin materials.
2. Biologics and Injectable Therapies
The rapid expansion of biologics and specialty drugs is accelerating demand for high-purity, low-extractable polyolefin grades. These materials offer enhanced compatibility with sensitive formulations, improved barrier properties, and sterilization stability.
3. Regulatory Compliance Requirements
Stringent global standards, including FDA and pharmacopeial guidelines, are driving the shift toward advanced primary packaging systems with improved moisture resistance, traceability, and safety performance.
Segment Analysis: Polyethylene Leads, Rigid Packaging Dominates
By Polyolefin Type
Polyethylene (PE) is expected to account for 50.4% of market share in 2026, driven by its versatility, chemical resistance, and compatibility with blow molding and film extrusion processes. PE is widely used for bottles, liners, and flexible laminates, supporting both cost efficiency and regulatory compliance.
By Packaging Format
Rigid packaging remains the dominant segment, particularly for tablets, capsules, and liquid medications, where consistent quality, barrier integrity, and tamper evidence are essential.
By End User
Branded pharmaceutical manufacturers are projected to hold 37.4% of market share in 2026, reflecting their focus on high-performance packaging, global regulatory compliance, and product differentiation. These companies prioritize validated supply chains and advanced packaging features such as child-resistant closures and serialization.
Regional Insights: Asia-Pacific Emerges as a Growth Hub
The global market is witnessing differentiated growth patterns across regions, with Asia-Pacific emerging as a key pharmaceutical manufacturing hub.
• India (6.2% CAGR): Growth is driven by strong generic drug production, export demand, and regulatory upgrades in packaging compliance.
• China (5.8% CAGR): Expansion is supported by increasing API production, regulatory enforcement, and modernization of pharmaceutical manufacturing.
• Brazil (5.3% CAGR): Rising domestic pharmaceutical consumption and public healthcare procurement are fueling demand.
• South Korea (4.9% CAGR): Growth is linked to biopharmaceutical exports and advanced manufacturing capabilities.
• Indonesia (4.7% CAGR): Increasing domestic production and healthcare investment are supporting steady expansion.
While emerging markets are driving volume growth, mature regions such as North America and Europe remain compliance-driven and replacement-oriented, focusing on upgrading packaging systems rather than expanding volume.
Competitive Landscape: Compliance and Integration Drive Advantage
The polyolefin pharmaceutical packaging market is moderately concentrated, with leading players including Amcor Plc, Gerresheimer AG, AptarGroup, Inc., West Pharmaceutical Services, Inc., and Constantia Flexibles Group GmbH. These companies maintain competitive advantage through:
• GMP-certified manufacturing facilities
• Validated pharmaceutical-grade material portfolios
• Integration across film extrusion, blow molding, and injection molding
• Strong relationships with pharmaceutical manufacturers
Regulatory qualification requirements create high entry barriers, as packaging materials must be validated within drug master files (DMFs), limiting supplier switching and stabilizing long-term supply relationships.
Innovation and Sustainability Trends
The market is transitioning toward advanced, high-purity polyolefins that offer improved barrier performance, reduced extractables, and enhanced drug stability. At the same time, sustainability is becoming a key focus area, with manufacturers developing recyclable mono-material structures and lightweight packaging solutions.
In August 2025, Constantia Flexibles announced a €100 million investment to expand its portfolio of mono-polyethylene and mono-polypropylene packaging solutions, reinforcing industry efforts to align performance with environmental objectives.
Why FMI: https://www.futuremarketinsights.com/why-fmi
Have a Look at Related Research Reports on the Packaging Domain:
High Airtight Storage Cabinets Market https://www.futuremarketinsights.com/reports/high-airtight-storage-cabinets-market
Edible Oil Cans Market https://www.futuremarketinsights.com/reports/edible-oil-cans-market
Multi Pocket Holders Market https://www.futuremarketinsights.com/reports/multi-pocket-holders-market
Cross Corner Industrial Bags Market https://www.futuremarketinsights.com/reports/cross-corner-industrial-bags-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.